Clinical Focus ›› 2026, Vol. 41 ›› Issue (3): 235-240.doi: 10.3969/j.issn.1004-583X.2026.03.007
Previous Articles Next Articles
Safety and efficacy of nafamostat mesylate as extracorporeal anticoagulation during continuous renal replacement therapy in uremic patients
- Department of Nephrology, Shijingshan Teaching Hospital of Capital Medical University, Beijing Shijingshan Hospital,Beijing 100043, China
-
Received:2025-10-15Online:2026-03-20Published:2026-03-27
CLC Number:
Cite this article
Shao Surong, Guo Yan. Safety and efficacy of nafamostat mesylate as extracorporeal anticoagulation during continuous renal replacement therapy in uremic patients[J]. Clinical Focus, 2026, 41(3): 235-240.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.lchc.cn/EN/10.3969/j.issn.1004-583X.2026.03.007
| 项目 | 观察组(n=29) | 对照组(n=29) | t/χ2值 | P值 |
|---|---|---|---|---|
| 年龄(岁) | 64.00±9.81 | 64.10±9.66 | -0.040 | 0.968 |
| 男性[例(%)] | 17(58.6) | 16(55.2) | -0.261 | 0.795 |
| APTT(s) | 27.17±1.64 | 28.27±3.00 | -1.742 | 0.089 |
| PLT(×109/L) | 165.38±51.75 | 167.10±64.27 | -1.113 | 0.911 |
| 血K(mmol/L) | 4.82±0.72 | 5.08±0.72 | -1.329 | 0.189 |
| ALT(U/L) | 19.83±7.53 | 18.48±10.38 | 0.565 | 0.575 |
| AST(U/L) | 20.41±7.72 | 19.55±9.01 | 2.661 | 0.060 |
| 原发病[例(%)] | ||||
| 糖尿病性肾病 | 10(34.48) | 11(37.93) | 0.075 | 0.785 |
| 慢性肾小球肾炎 | 12(41.38) | 10(34.48) | 0.293 | 0.588 |
| 高血压性肾病 | 4(13.79) | 6(20.69) | 0.483 | 0.487 |
| 其他 | 3(10.35) | 2(6.90) | 0.345 | 0.557 |
Tab.1 Baseline data between groups
| 项目 | 观察组(n=29) | 对照组(n=29) | t/χ2值 | P值 |
|---|---|---|---|---|
| 年龄(岁) | 64.00±9.81 | 64.10±9.66 | -0.040 | 0.968 |
| 男性[例(%)] | 17(58.6) | 16(55.2) | -0.261 | 0.795 |
| APTT(s) | 27.17±1.64 | 28.27±3.00 | -1.742 | 0.089 |
| PLT(×109/L) | 165.38±51.75 | 167.10±64.27 | -1.113 | 0.911 |
| 血K(mmol/L) | 4.82±0.72 | 5.08±0.72 | -1.329 | 0.189 |
| ALT(U/L) | 19.83±7.53 | 18.48±10.38 | 0.565 | 0.575 |
| AST(U/L) | 20.41±7.72 | 19.55±9.01 | 2.661 | 0.060 |
| 原发病[例(%)] | ||||
| 糖尿病性肾病 | 10(34.48) | 11(37.93) | 0.075 | 0.785 |
| 慢性肾小球肾炎 | 12(41.38) | 10(34.48) | 0.293 | 0.588 |
| 高血压性肾病 | 4(13.79) | 6(20.69) | 0.483 | 0.487 |
| 其他 | 3(10.35) | 2(6.90) | 0.345 | 0.557 |
| 组别 | 例数 | 0级 | Ⅰ级 | Ⅱ级 | Ⅲ级 | 抗凝有效率 |
|---|---|---|---|---|---|---|
| 观察组 | 29 | 25(86.2) | 2(6.9) | 2(6.9) | 0 | 27(93.1) |
| 对照组 | 29 | 25(86.2) | 3(10.3) | 1(3.5) | 0 | 28(96.6) |
| χ2值 | 0.352 | |||||
| P值 | 0.553 |
Tab.2 Effective rate of anticoagulation between groups (n, %)
| 组别 | 例数 | 0级 | Ⅰ级 | Ⅱ级 | Ⅲ级 | 抗凝有效率 |
|---|---|---|---|---|---|---|
| 观察组 | 29 | 25(86.2) | 2(6.9) | 2(6.9) | 0 | 27(93.1) |
| 对照组 | 29 | 25(86.2) | 3(10.3) | 1(3.5) | 0 | 28(96.6) |
| χ2值 | 0.352 | |||||
| P值 | 0.553 |
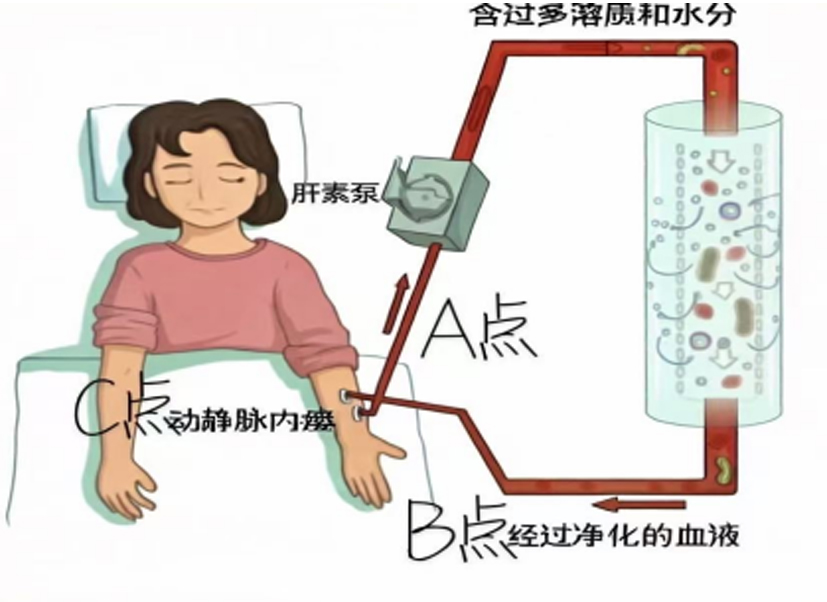
| 变量 | A采血点 | C采血点血滤结束15 min | F值 | P值 | ||
|---|---|---|---|---|---|---|
| CRRT 0 h | CRRT 4 h | CRRT结束 | ||||
| APTT(s) | 27.16±1.64 | 27.77±1.93 | 28.85±2.44 | 27.73±2.74 | 2.901 | 0.061 |
Tab.3 Comparison of APTT at different time points at Site A and 15 minutes after the end of CRRT at Site C in the observation group(n=29)
| 变量 | A采血点 | C采血点血滤结束15 min | F值 | P值 | ||
|---|---|---|---|---|---|---|
| CRRT 0 h | CRRT 4 h | CRRT结束 | ||||
| APTT(s) | 27.16±1.64 | 27.77±1.93 | 28.85±2.44 | 27.73±2.74 | 2.901 | 0.061 |
| 变量 | A采血点CRRT0 h | C采血点CRRT结束15 min | t值 | P值 |
|---|---|---|---|---|
| PLT(×109/L) | 165.38±51.75 | 162.28±45.85 | 0.242 | 0.810 |
| 血K(mmol/L) | 4.82±0.72 | 4.32±0.52 | 3.070 | 0.003 |
| ALT(U/L) | 19.83±7.53 | 18.94±6.71 | 3.995 | 0.078 |
| AST(U/L) | 20.41±7.72 | 19.72±7.38 | 2.684 | 0.354 |
Tab.4 Indicators of observation group at A and C time points (n=29)
| 变量 | A采血点CRRT0 h | C采血点CRRT结束15 min | t值 | P值 |
|---|---|---|---|---|
| PLT(×109/L) | 165.38±51.75 | 162.28±45.85 | 0.242 | 0.810 |
| 血K(mmol/L) | 4.82±0.72 | 4.32±0.52 | 3.070 | 0.003 |
| ALT(U/L) | 19.83±7.53 | 18.94±6.71 | 3.995 | 0.078 |
| AST(U/L) | 20.41±7.72 | 19.72±7.38 | 2.684 | 0.354 |
| 组别 | 例数 | APTT(A点) | ||
|---|---|---|---|---|
| CRRT 0 h | CRRT 4 h | CRRT结束时 | ||
| 观察组 | 29 | 27.17±1.64 | 27.77±1.93 | 28.85±2.44 |
| 对照组 | 29 | 28.27±3.00 | 78.78±11.45 | 75.77±9.86 |
| t值 | -1.742 | -23.663 | -24.883 | |
| P值 | 0.087 | <0.001 | <0.001 | |
Tab.5 APTT Parameters at site A across time intervals between groups during hemofiltration (s)
| 组别 | 例数 | APTT(A点) | ||
|---|---|---|---|---|
| CRRT 0 h | CRRT 4 h | CRRT结束时 | ||
| 观察组 | 29 | 27.17±1.64 | 27.77±1.93 | 28.85±2.44 |
| 对照组 | 29 | 28.27±3.00 | 78.78±11.45 | 75.77±9.86 |
| t值 | -1.742 | -23.663 | -24.883 | |
| P值 | 0.087 | <0.001 | <0.001 | |
| 组别 | 例数 | APTT(B点) | ||
|---|---|---|---|---|
| CRRT 0 h | CRRT 4 h | CRRT结束时 | ||
| 观察组 | 29 | 80.58±9.37 | 85.69±7.93 | 77.08±10.88 |
| 对照组 | 29 | 81.39±9.78 | 83.07±10.10 | 76.72±9.49 |
| t值 | -0.321 | 1.098 | 0.134 | |
| P值 | 0.750 | 0.277 | 0.894 | |
Tab.6 APTT parameters at site B across time intervals between groups during hemofiltration (s)
| 组别 | 例数 | APTT(B点) | ||
|---|---|---|---|---|
| CRRT 0 h | CRRT 4 h | CRRT结束时 | ||
| 观察组 | 29 | 80.58±9.37 | 85.69±7.93 | 77.08±10.88 |
| 对照组 | 29 | 81.39±9.78 | 83.07±10.10 | 76.72±9.49 |
| t值 | -0.321 | 1.098 | 0.134 | |
| P值 | 0.750 | 0.277 | 0.894 | |
| 组别 | 例数 | C点,CRRT结束15 min | ||||
|---|---|---|---|---|---|---|
| APTT | PLT | 血K | ALT | AST | ||
| 观察组 | 29 | 27.73±2.74 | 162.28±45.85 | 4.32±0.52 | 13.24±4.71 | 15.72±5.38 |
| 对照组 | 29 | 37.32±6.30 | 133.69±40.63 | 4.81±0.49 | 12.45±4.00 | 14.62±4.48 |
| t值 | -7.514 | 2.513 | 4.185 | 0.691 | 3.154 | |
| P值 | <0.001 | 0.015 | 0.351 | 0.492 | 0.235 | |
Tab.7 APTT parameters at site C across time intervals between groups during hemofiltration (s)
| 组别 | 例数 | C点,CRRT结束15 min | ||||
|---|---|---|---|---|---|---|
| APTT | PLT | 血K | ALT | AST | ||
| 观察组 | 29 | 27.73±2.74 | 162.28±45.85 | 4.32±0.52 | 13.24±4.71 | 15.72±5.38 |
| 对照组 | 29 | 37.32±6.30 | 133.69±40.63 | 4.81±0.49 | 12.45±4.00 | 14.62±4.48 |
| t值 | -7.514 | 2.513 | 4.185 | 0.691 | 3.154 | |
| P值 | <0.001 | 0.015 | 0.351 | 0.492 | 0.235 | |
| 组别 | 例数 | 消化道出血 | 牙龈出血 | 血尿 | 眼底出血 | 出血发生率 |
|---|---|---|---|---|---|---|
| 观察组 | 29 | 0 | 1(3.45) | 0 | 1(3.45) | 2(6.90) |
| 对照组 | 29 | 2(6.89) | 3(10.35) | 2(6.89) | 2(6.89) | 9(31.02) |
| χ2值 | 5.402 | |||||
| P值 | 0.041 |
Tab.8 Incidence of bleeding between groups (n, %)
| 组别 | 例数 | 消化道出血 | 牙龈出血 | 血尿 | 眼底出血 | 出血发生率 |
|---|---|---|---|---|---|---|
| 观察组 | 29 | 0 | 1(3.45) | 0 | 1(3.45) | 2(6.90) |
| 对照组 | 29 | 2(6.89) | 3(10.35) | 2(6.89) | 2(6.89) | 9(31.02) |
| χ2值 | 5.402 | |||||
| P值 | 0.041 |
| [1] | Zhang H, Li G, Yu X, et al. Progression of vascular calcification and clinical outcomes in patients re ceiving maintenance dialysis[J]. JAMA Netw Open, 2023, 6 (5):e2310909.doi: 10.1001/jamanetworkopen.2023.10909. |
| [2] | Gaudry S, Hajage D, Schortgen F, et al. Initiation strategies for renal-replacement therapy in the intensive care unit[J]. NewEngl J Med, 2016, 375( 2): 122-33.doi:10.1056/NEJMoa1603017. |
| [3] | 周芯蕊, 沈清. 萘莫司他抗凝在连续性肾脏替代治疗中应用的研究进展[J]. 中国医药, 2024, 19(8): 1263-1266.doi:10.3760./j.issn.1633-4777.2024.08.031. |
| [4] | Legrand M, Tolwani A. Anticoagulation strategies in continuous renal replacement therapy[J]. Semin Dial, 2021, 34(6): 416-422.doi: 10.1111/sdi.12959. |
| [5] | 唐小琰, 张凌, 付平. 局部枸橼酸抗凝在间歇性血液透析中的应用进展[J]. 中国血液净化, 2021, 20(6): 387-390.doi:10.3969/j.issn.1671-4091.2021.06.007. |
| [6] |
Lin Y, Shao Y, Liu Y, et al. Efficacy and safety of nafamostat mesilate anticoagulation in blood purification treatment of critically ill patients: A systematic review and meta-analysis[J]. Ren Fail, 2022, 44(1): 1263-1279. doi: 10.1080/0886022X.2022.2105233.
pmid: 35930302 |
| [7] |
Arimura T, Abe M, Shiga H, et al. Clinical study of blood purification therapy in critical care in Japan: Results from the survey research of the Japan Society for Blood Purification in Critical Care in 2013[J]. J Artif Organs, 2017, 20(3): 244-251. doi: 10.1007/s10047-017-0968-3.
pmid: 28600615 |
| [8] |
Lang YH, Zheng Y, Qi BC, et al. Anticoagulation with nafamostat mesilate during extracorporeal life support[J]. Int J Cardiol, 2022, 366: 71-79. doi:5.1016/j.ijcard.2022.07.022.
pmid: 35850387 |
| [9] | 胡家昌, 谢烨卿, 沈波, 等. 甲磺酸萘莫司他的血液净化抗凝应用专家共识[J]. 上海医学, 2024, 47(3): 129-144.doi:10.19842/j.cnki.issn.0253-9934.2024.03.001. |
| [10] |
Konishi K, SInoue S, Kawaguchi M. Evaluation of the pharmacokinetics of nafamostat mesylate during contionuous renal replacement therapy[J]. OJEM, 2022, 10(4):157-167.doi.org/10.4236/ojem.2022.104014.
doi: 10.4236/ojem.2022.104014 URL |
| [11] | Miyatake Y, Makino S, Kubota K, et al. Association between intra-circuit activated clotting time and incidence of bleeding complications during continuous renal replacement therapy using nafamostat mesilate: A retrospective pilot observational study[J]. Kobe J Med Sci, 2017, 63(1):E30-E36. |
| [12] |
Fuse I, Higuchi W, Toba K, Aizawa Y. Inhibitory mechanism of human platelet aggregation by nafamostat mesilate[J]. Platelets, 1999, 10(4):212-218. doi: 10.1080/09537109976040.
pmid: 16801094 |
| [13] | Iwama H, Watanabe K, Obara S. In vitro relationship between the blood nafamostat concentration and activated coagulation time[J]. Can J Anaesth, 2003, 50(6):621-622. doi: 10.1007/BF03018658. |
| [14] |
Pelletier R, Nagge J, Gamble JM. Variationin bleeding risk estimates among online calculators: Cross-sectional study of Apps used by and for patients with atrial fibrillation[J]. Can Fam Physician, 2022, 68(4):e127-e135.doi:10.46747/cfp.6804e127.
pmid: 35418403 |
| [15] | Zhang W, Bai M, Zhang L, et al. Developmentand external validation of amodel for predicting sufficient filter life spanin anticoagulation-free continuous renal replacement therapy patients[J]. Blood Purif, 2022, 51(8):668-678.doi:10.1159/000519409. |
| [1] | Zhang Yanping, Li Ke, Zhang Zhuang. Safety of an early initiation of anticoagulant therapy for atrial fibrillation associated acute cerebral infarction [J]. Clinical Focus, 2026, 41(2): 116-121. |
| [2] | Su Rui, Wang Cunkai, Wang Dingxin, Cai Conghui, Zhang Jian, Hou Hongtao, Bai Yun. Efficacy and safety of anticoagulant therapy in patients with cirrhosis: A meta-analysis [J]. Clinical Focus, 2025, 40(4): 293-303. |
| [3] | Cui Zhijun, Ma Donghong, Chen Xiufeng. Impact of high flow dialysis on sleep quality in maintenance hemodialysis patients and analysis of related factors [J]. Clinical Focus, 2025, 40(12): 1082-1086. |
| [4] | Guo Xiaocui, Yu Xiaojuan, Shen Xia, Ye Shuiying, Zhou Dongchi, Lai Bihong. Effect of a multidisciplinary intervention on the quality of life in maintenance hemodialysis patients [J]. Clinical Focus, 2024, 39(9): 803-807. |
| [5] | Yu Jingyi, Si Yuanguo, Lan Cuixia, Xing Jiaxuan. Safety and efficacy of rivaroxaban in the treatment of discharged COVID-19 patients: A meta-analysis [J]. Clinical Focus, 2024, 39(6): 485-493. |
| [6] | Wang Yihan, Qin Xuyan, Han Xuanze, Wang Yingjie, Gao Feifei, Chen Chunhong, Zhang Lingnan, Zhang Fang. Efficacy and safety of rivaroxaban therapy in elderly patients with non-valvular atrial fibrillation who had a HASBLED score≥3 [J]. Clinical Focus, 2024, 39(2): 121-124. |
| [7] | Tao Yang, Pan Qingquan, Li Yan. Evaluation of hemoperfusion and continuous renal replacement therapy in paraquat poisoning patients complicated with MODS [J]. Clinical Focus, 2023, 38(4): 330-334. |
| [8] | Zhou Huixian, Zhou Yaqing, Guo Ganlin, Cui Wei. Therapy on left ventricular thrombus:Systematic review and meta-analysis [J]. Clinical Focus, 2023, 38(2): 101-110. |
| [9] | Dai Jing, Chen Huaqian. Wunderlich syndrome in a patient on haemodialysis with a review of the literature [J]. Clinical Focus, 2023, 38(12): 1107-1111. |
| [10] | Li Wenzhe, Shang Jinchun, Li Chunmei, Tian Fen, Li Jun, Cui Li, Xing Guangqun. Risk factors of cerebral hemorrhage in uremic patients on regular hemodialysis [J]. Clinical Focus, 2022, 37(1): 20-25. |
| [11] | Li Wenzhe, Shang Jinchun, Li Chunmei, Li Jun, Tian Fen, Cui Li, Chen Yipeng, Zhang Xiaofan, Xing Guangqun. Effect of sodium citrate anticoagulation in continuous renal replacement therapy on uremic patients with cerebral hemorrhage [J]. Clinical Focus, 2021, 36(5): 425-431. |
| [12] | Huang Junyue, Ma Zhigang, Li Yingping, Xue Rong, Huang Wenhui, Jing Xiaojiang. Effects of high-flux hemodialysis combined with L-carnitine treatment on lipid metabolism in uremic patients [J]. Clinical Focus, 2019, 34(3): 253-256. |
| [13] | Xu Bingdong, Mai Hongcheng, Wu Zhengdong, Zhu Peizhi, Liang Yubin, Zhang Yusheng. Progress in diagnosis and treatment of embolic stroke of undetermined source [J]. Clinical Focus, 2018, 33(3): 185-188,193. |
| [14] | Ding Hong;Pan Jieping;Zhu Jinhua;Tian Ying;Song Lei;Shen Minning. Five cases of scleroderma renal crisis combined with literature review [J]. Clinical Focus, 2015, 30(9): 1013-1018. |
| [15] | Yu Qin;Xing Guangqun;Zhou Quan. Effects of neutrophil extracellular traps and contact system in uremic patients with acute coronary syndrome [J]. Clinical Focus, 2015, 30(5): 540-543544. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||