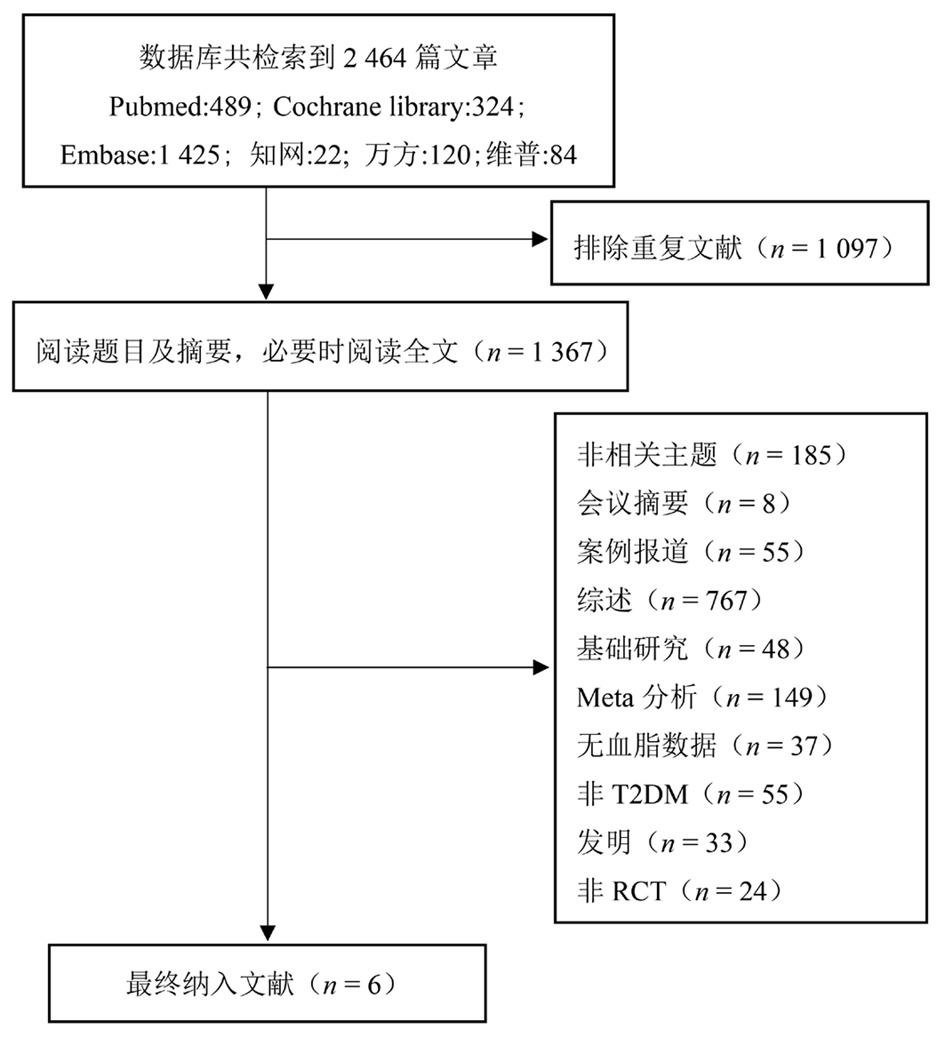
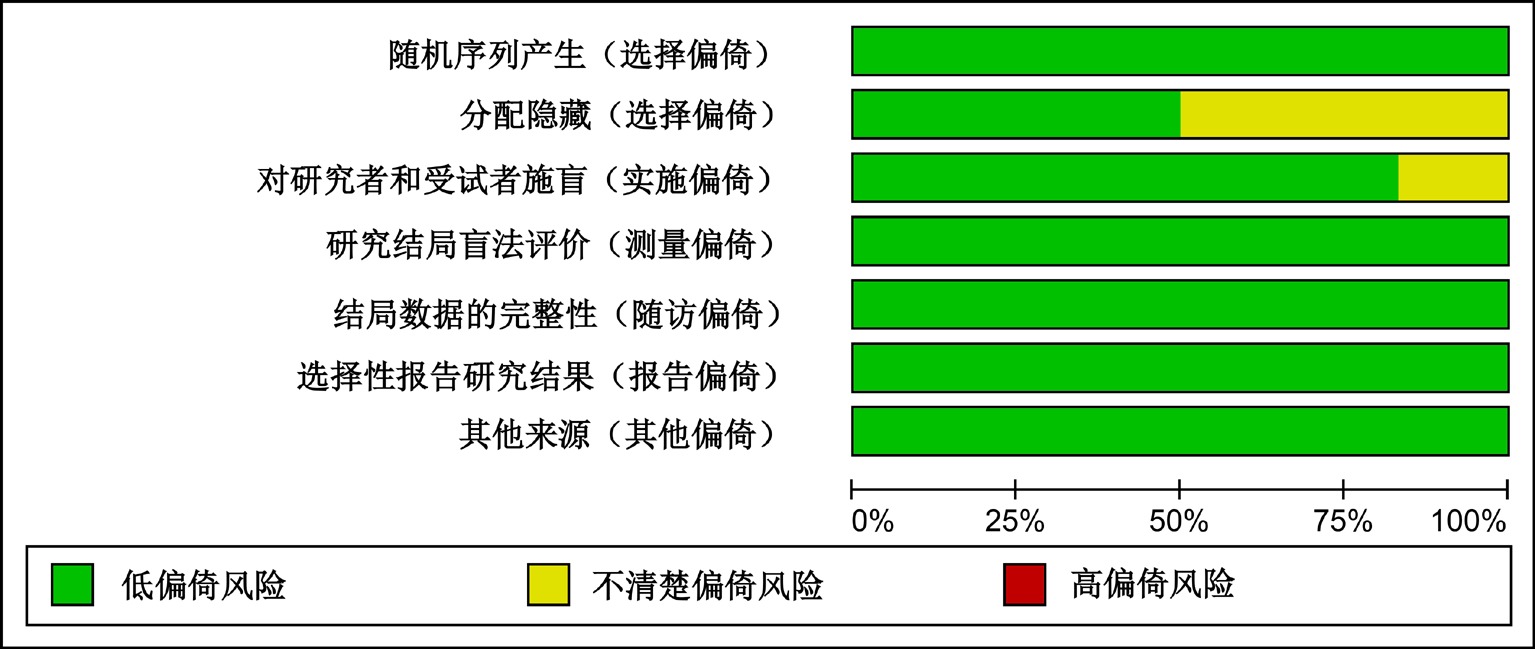
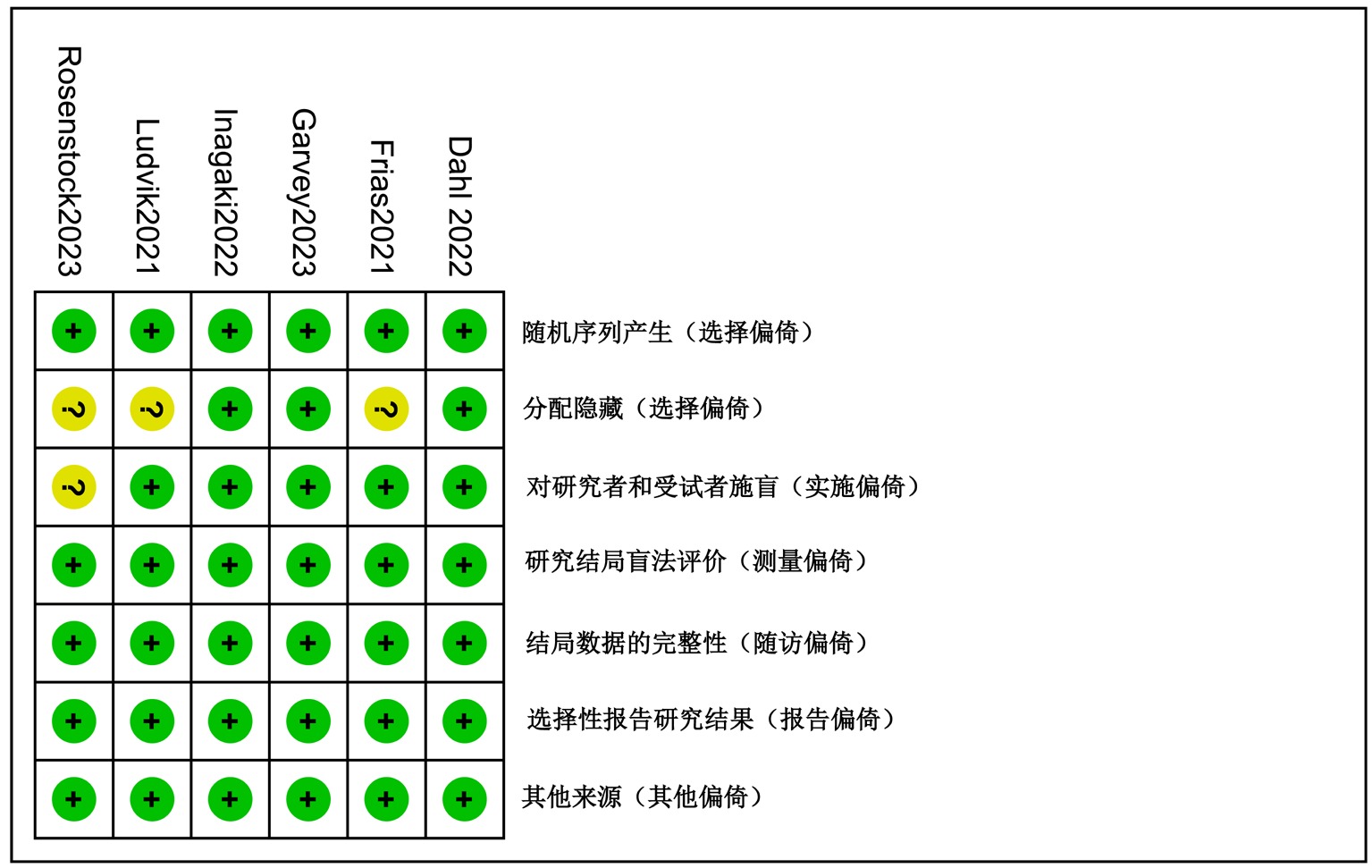
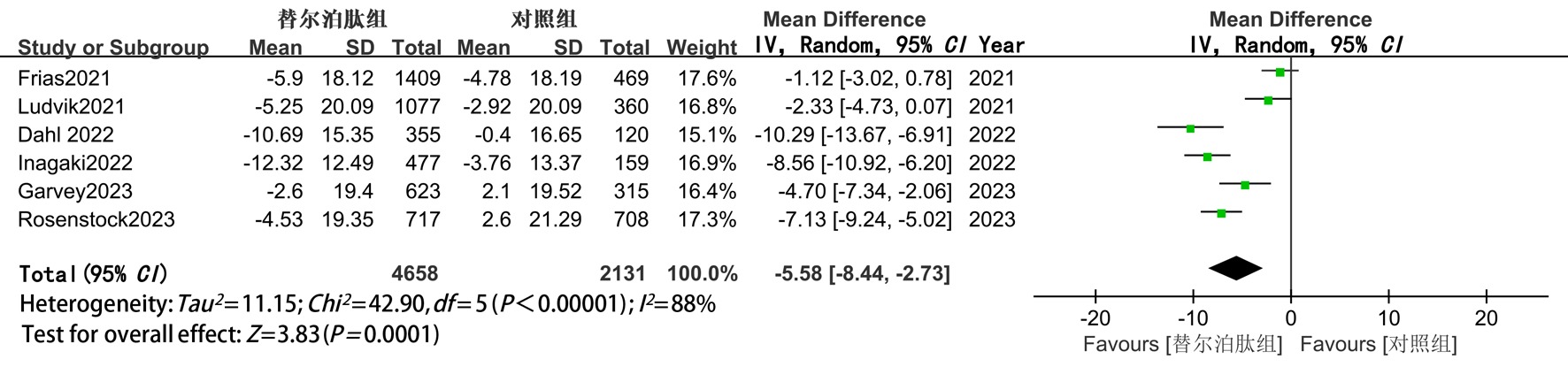
Clinical Focus ›› 2025, Vol. 40 ›› Issue (12): 1061-1067.doi: 10.3969/j.issn.1004-583X.2025.12.001
Effects of tirzepatide on lipid profiles in type 2 diabetes mellitus: A meta-analysis
- 1. Department of Clinical Nutrition,the Fourth Affiliated Hospital of Guangxi Medical University, Liuzhou 545000,China
2. Department of Emergency Medicine,Liuzhou People's Hospital Affiliated to Guangxi Medical University,Liuzhou 545006,China
-
Received:2025-09-05Online:2025-12-20Published:2025-12-30 -
Contact:Zhang Weijian,Email: cheungweijian@163.com
CLC Number:
Cite this article
Xie Feifei, Zhang Weijian. Effects of tirzepatide on lipid profiles in type 2 diabetes mellitus: A meta-analysis[J]. Clinical Focus, 2025, 40(12): 1061-1067.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.lchc.cn/EN/10.3969/j.issn.1004-583X.2025.12.001
Tab.1 Baseline characteristics and quality assessment of included studies
| 纳入研究 | 发表年份 | 干预时间 (周) | 干预措施 | 研究人数 | 年龄 (岁) | HbA1c (%) | 体重 (kg) | 质量等级 |
|---|---|---|---|---|---|---|---|---|
| Frias等[ | 2021 | 40 | Tirzepatide 5 mg | 470 | 56.3±10.0 | 8.32±1.08 | 92.5±21.8 | B |
| Tirzepatide 10 mg | 469 | 57.2±10.5 | 8.30±1.02 | 94.8±22.7 | ||||
| Tirzepatide 15 mg | 470 | 55.9±10.4 | 8.26±1.00 | 93.8±21.8 | ||||
| 司美格鲁肽1 mg | 469 | 56.9±10.8 | 8.25±1.01 | 93.7±21.1 | ||||
| Ludvik等[ | 2021 | 52 | Tirzepatide 5 mg | 358 | 57.2±10.1 | 8.17±0.89 | 94.4±18.9 | B |
| Tirzepatide 10 mg | 360 | 57.4±9.7 | 8.18±0.89 | 93.8±19.8 | ||||
| Tirzepatide 15 mg | 359 | 57.5±10.2 | 8.21±0.94 | 94.9±21.0 | ||||
| 德谷胰岛素 | 360 | 57.5±10.1 | 8.12±0.94 | 94.0±20.6 | ||||
| Dahl等[ | 2022 | 40 | Tirzepatide 5 mg | 116 | 61.5±9.8 | 8.30±0.88 | 95.8±19.8 | A |
| Tirzepatide 10 mg | 119 | 60.4±10.2 | 8.36±0.83 | 94.5±22.2 | ||||
| Tirzepatide 15 mg | 120 | 60.5±10.0 | 8.23±0.86 | 96.3±22.8 | ||||
| 安慰剂 | 120 | 60.0±9.6 | 8.37±0.84 | 94.1±21.8 | ||||
| Inagaki等[ | 2022 | 52 | Tirzepatide 5 mg | 159 | 56.8±10.1 | 8.18±0.88 | 78.5±16.3 | A |
| Tirzepatide 10 mg | 158 | 56.2±10.3 | 8.19±0.86 | 78.9±13.7 | ||||
| Tirzepatide 15 mg | 160 | 56.0±10.7 | 8.19±0.89 | 78.9±14.3 | ||||
| 度拉鲁肽0.75 mg | 159 | 57.5±10.2 | 8.15±0.86 | 76.5±13.2 | ||||
| Garvey等[ | 2023 | 72 | Tirzepatide 10 mg | 312 | 54.3±10.7 | 8.00±0.84 | 100.9±20.9 | A |
| Tirzepatide 15 mg | 311 | 53.6±10.6 | 8.07±0.99 | 99.6±20.1 | ||||
| 安慰剂 | 315 | 54.7±10.5 | 7.98±0.84 | 101.7±22.3 | ||||
| Rosenstock等[ | 2023 | 52 | Tirzepatide 5 mg | 243 | 58.0±10.2 | 8.89±0.97 | 91.7±17.9 | B |
| Tirzepatide 10 mg | 238 | 59.6±9.4 | 8.78±0.98 | 89.1±18.8 | ||||
| Tirzepatide 15 mg | 236 | 58.2±9.6 | 8.74±1.01 | 91.2±18.7 | ||||
| 赖脯胰岛素 | 708 | 59.0±9.7 | 8.80±0.96 | 90.3±17.7 |
Tab.1 Baseline characteristics and quality assessment of included studies
| 纳入研究 | 发表年份 | 干预时间 (周) | 干预措施 | 研究人数 | 年龄 (岁) | HbA1c (%) | 体重 (kg) | 质量等级 |
|---|---|---|---|---|---|---|---|---|
| Frias等[ | 2021 | 40 | Tirzepatide 5 mg | 470 | 56.3±10.0 | 8.32±1.08 | 92.5±21.8 | B |
| Tirzepatide 10 mg | 469 | 57.2±10.5 | 8.30±1.02 | 94.8±22.7 | ||||
| Tirzepatide 15 mg | 470 | 55.9±10.4 | 8.26±1.00 | 93.8±21.8 | ||||
| 司美格鲁肽1 mg | 469 | 56.9±10.8 | 8.25±1.01 | 93.7±21.1 | ||||
| Ludvik等[ | 2021 | 52 | Tirzepatide 5 mg | 358 | 57.2±10.1 | 8.17±0.89 | 94.4±18.9 | B |
| Tirzepatide 10 mg | 360 | 57.4±9.7 | 8.18±0.89 | 93.8±19.8 | ||||
| Tirzepatide 15 mg | 359 | 57.5±10.2 | 8.21±0.94 | 94.9±21.0 | ||||
| 德谷胰岛素 | 360 | 57.5±10.1 | 8.12±0.94 | 94.0±20.6 | ||||
| Dahl等[ | 2022 | 40 | Tirzepatide 5 mg | 116 | 61.5±9.8 | 8.30±0.88 | 95.8±19.8 | A |
| Tirzepatide 10 mg | 119 | 60.4±10.2 | 8.36±0.83 | 94.5±22.2 | ||||
| Tirzepatide 15 mg | 120 | 60.5±10.0 | 8.23±0.86 | 96.3±22.8 | ||||
| 安慰剂 | 120 | 60.0±9.6 | 8.37±0.84 | 94.1±21.8 | ||||
| Inagaki等[ | 2022 | 52 | Tirzepatide 5 mg | 159 | 56.8±10.1 | 8.18±0.88 | 78.5±16.3 | A |
| Tirzepatide 10 mg | 158 | 56.2±10.3 | 8.19±0.86 | 78.9±13.7 | ||||
| Tirzepatide 15 mg | 160 | 56.0±10.7 | 8.19±0.89 | 78.9±14.3 | ||||
| 度拉鲁肽0.75 mg | 159 | 57.5±10.2 | 8.15±0.86 | 76.5±13.2 | ||||
| Garvey等[ | 2023 | 72 | Tirzepatide 10 mg | 312 | 54.3±10.7 | 8.00±0.84 | 100.9±20.9 | A |
| Tirzepatide 15 mg | 311 | 53.6±10.6 | 8.07±0.99 | 99.6±20.1 | ||||
| 安慰剂 | 315 | 54.7±10.5 | 7.98±0.84 | 101.7±22.3 | ||||
| Rosenstock等[ | 2023 | 52 | Tirzepatide 5 mg | 243 | 58.0±10.2 | 8.89±0.97 | 91.7±17.9 | B |
| Tirzepatide 10 mg | 238 | 59.6±9.4 | 8.78±0.98 | 89.1±18.8 | ||||
| Tirzepatide 15 mg | 236 | 58.2±9.6 | 8.74±1.01 | 91.2±18.7 | ||||
| 赖脯胰岛素 | 708 | 59.0±9.7 | 8.80±0.96 | 90.3±17.7 |
Tab.2 Subgroup meta analysis results of different dose of tirzepatide
| 指标 | 模型 | 总体 | 替尔泊肽剂量亚组分析 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MD(95%CI) | P值 | 5 mg vs对照组 | 10 mg vs对照组 | 15 mg vs对照组 | |||||
| MD(95%CI) | P值 | MD(95%CI) | P值 | MD(95%CI) | P值 | ||||
| TC | 随机 | -5.59(-7.33,-3.85) | <0.05 | -4.65(-7.78,-1.51) | <0.05 | -5.40(-7.97,-2.83) | <0.05 | -6.58(-10.24,-2.91) | <0.05 |
| TG | 随机 | -15.94(-19.11,-12.77) | <0.05 | -9.73(-14.57,-4.89) | <0.05 | -16.81(-20.42,-13.20) | <0.05 | -19.89(-25.16,-14.62) | <0.05 |
| HDL-C | 随机 | 4.19(2.71,5.67) | <0.05 | 2.13(0.19,4.08) | <0.05 | 4.62(2.32,6.93) | <0.05 | 5.49(2.73,8.26) | <0.05 |
| LDL-C | 随机 | -6.49(-9.05,-3.93) | <0.05 | -5.98(-9.55,-2.41) | <0.05 | -5.79(-9.92,-1.66) | <0.05 | -7.58(-13.35,-1.80) | <0.05 |
| VLDL-C | 随机 | -15.74(-18.87,-12.60) | <0.05 | -9.62(-14.41,-4.83) | <0.05 | -16.59(-20.16,-13.01) | <0.05 | -19.57(-24.73,-14.41) | <0.05 |
Tab.2 Subgroup meta analysis results of different dose of tirzepatide
| 指标 | 模型 | 总体 | 替尔泊肽剂量亚组分析 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MD(95%CI) | P值 | 5 mg vs对照组 | 10 mg vs对照组 | 15 mg vs对照组 | |||||
| MD(95%CI) | P值 | MD(95%CI) | P值 | MD(95%CI) | P值 | ||||
| TC | 随机 | -5.59(-7.33,-3.85) | <0.05 | -4.65(-7.78,-1.51) | <0.05 | -5.40(-7.97,-2.83) | <0.05 | -6.58(-10.24,-2.91) | <0.05 |
| TG | 随机 | -15.94(-19.11,-12.77) | <0.05 | -9.73(-14.57,-4.89) | <0.05 | -16.81(-20.42,-13.20) | <0.05 | -19.89(-25.16,-14.62) | <0.05 |
| HDL-C | 随机 | 4.19(2.71,5.67) | <0.05 | 2.13(0.19,4.08) | <0.05 | 4.62(2.32,6.93) | <0.05 | 5.49(2.73,8.26) | <0.05 |
| LDL-C | 随机 | -6.49(-9.05,-3.93) | <0.05 | -5.98(-9.55,-2.41) | <0.05 | -5.79(-9.92,-1.66) | <0.05 | -7.58(-13.35,-1.80) | <0.05 |
| VLDL-C | 随机 | -15.74(-18.87,-12.60) | <0.05 | -9.62(-14.41,-4.83) | <0.05 | -16.59(-20.16,-13.01) | <0.05 | -19.57(-24.73,-14.41) | <0.05 |
Tab.3 Subgroup meta analysis results of different control drugs
| 指标 | 模型 | 总体 | 不同对照药物亚组分析 | ||||
|---|---|---|---|---|---|---|---|
| MD(95%CI) | P值 | 替尔泊肽vs GLP-1RA或胰岛素 | 替尔泊肽vs安慰剂 | ||||
| MD(95%CI) | P值 | MD(95%CI) | P值 | ||||
| TC | 随机 | -5.58(-8.4,-2.73) | <0.05 | -4.76(-8.36,-1.17) | <0.05 | -7.39(-12.87,-1.92) | <0.05 |
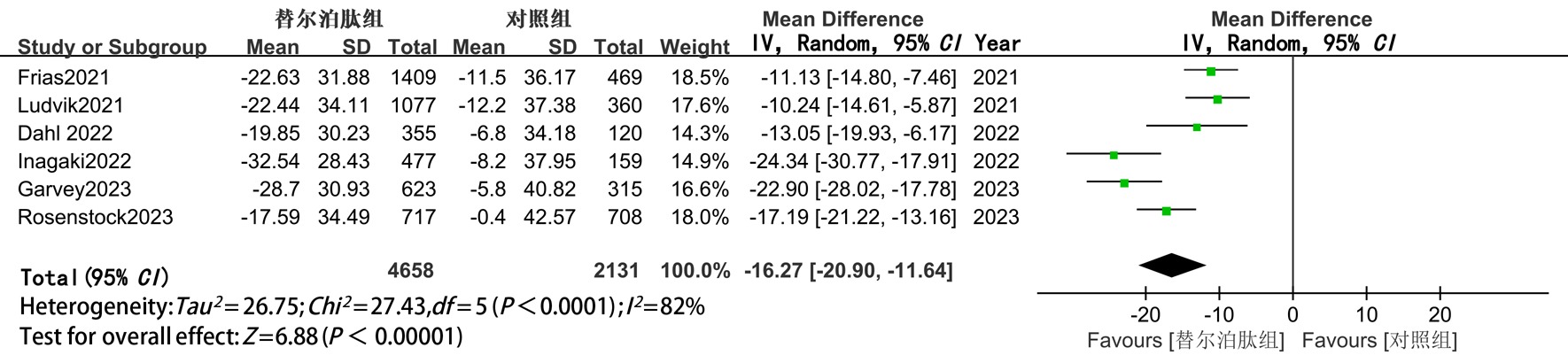
| TG | 随机 | -16.27(-20.90,-11.64) | <0.05 | -15.31(-20.71,-9.92) | <0.05 | -18.25(-27.89,-8.62) | <0.05 |
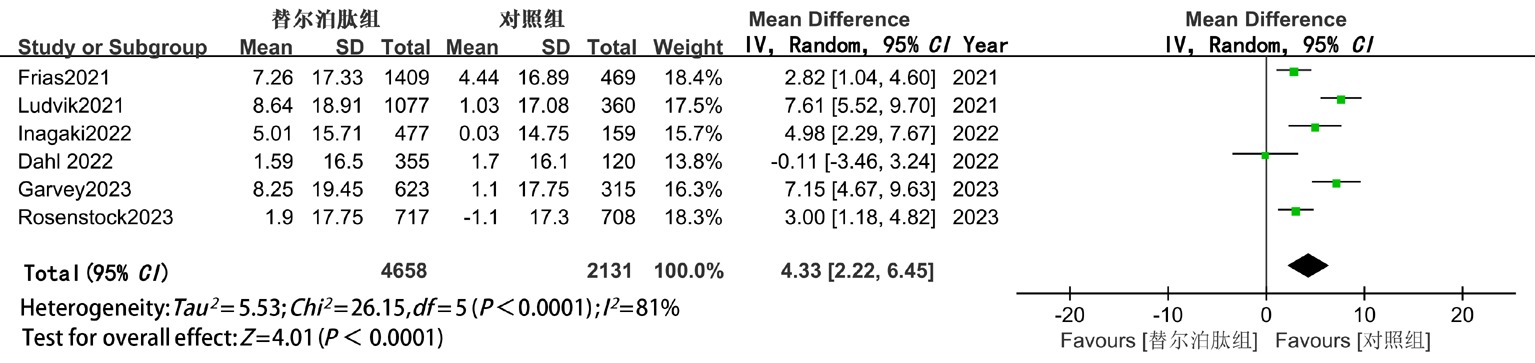
| HDL-C | 随机 | 4.33(2.22,6.45) | <0.05 | 4.54(2.28,6.80) | <0.05 | 3.61(-3.50,10.72) | >0.05 |
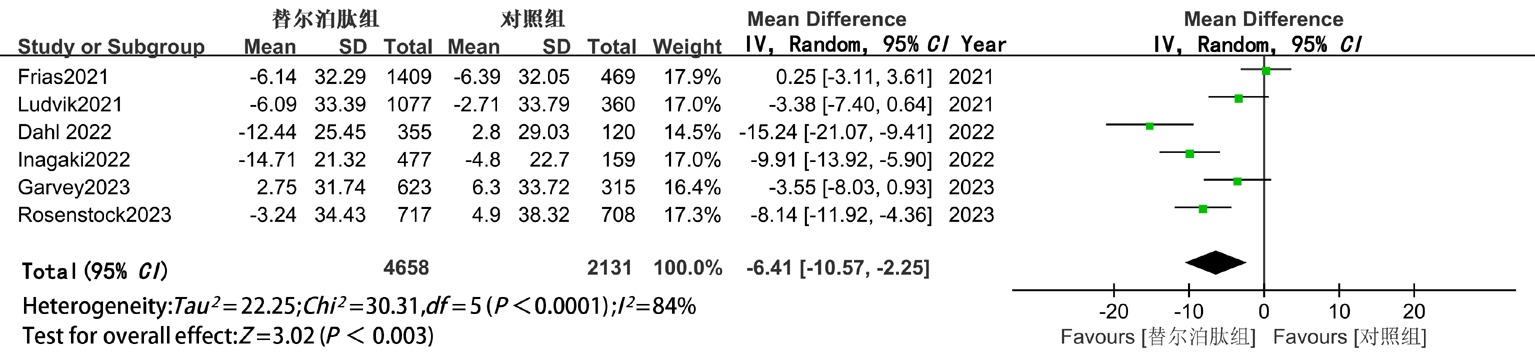
| LDL-C | 随机 | -6.41(-10.57,-2.25) | <0.05 | -5.23(-9.91,-0.55) | <0.05 | -9.24(-20.69,2.21) | >0.05 |
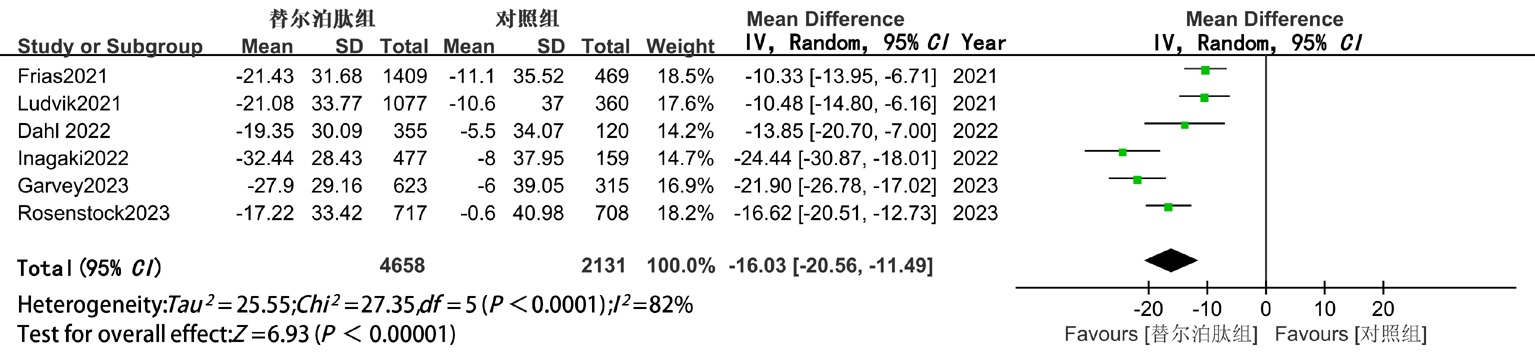
| VLDL-C | 随机 | -16.03(-20.56,-11.49) | <0.05 | -15.04(-20.47,-9.60) | <0.05 | -18.25(-26.10,-10.39) | <0.05 |
Tab.3 Subgroup meta analysis results of different control drugs
| 指标 | 模型 | 总体 | 不同对照药物亚组分析 | ||||
|---|---|---|---|---|---|---|---|
| MD(95%CI) | P值 | 替尔泊肽vs GLP-1RA或胰岛素 | 替尔泊肽vs安慰剂 | ||||
| MD(95%CI) | P值 | MD(95%CI) | P值 | ||||
| TC | 随机 | -5.58(-8.4,-2.73) | <0.05 | -4.76(-8.36,-1.17) | <0.05 | -7.39(-12.87,-1.92) | <0.05 |
| TG | 随机 | -16.27(-20.90,-11.64) | <0.05 | -15.31(-20.71,-9.92) | <0.05 | -18.25(-27.89,-8.62) | <0.05 |
| HDL-C | 随机 | 4.33(2.22,6.45) | <0.05 | 4.54(2.28,6.80) | <0.05 | 3.61(-3.50,10.72) | >0.05 |
| LDL-C | 随机 | -6.41(-10.57,-2.25) | <0.05 | -5.23(-9.91,-0.55) | <0.05 | -9.24(-20.69,2.21) | >0.05 |
| VLDL-C | 随机 | -16.03(-20.56,-11.49) | <0.05 | -15.04(-20.47,-9.60) | <0.05 | -18.25(-26.10,-10.39) | <0.05 |
| [1] | 中华医学会糖尿病学分会. 中国糖尿病防治指南(2024版)[J]. 中华糖尿病杂志, 2025, 17(1):16-139. doi:10.3760/cma.j.cn115791-20241203-00705. |
| [2] | Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes[J]. N Engl J Med, 2021, 385(6):503-515. doi:10.1056/NEJMoa2107519. |
| [3] |
Aronne LJ, Sattar N, Horn DB, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: The SURMOUNT-4 randomized clinical trial[J]. JAMA, 2024, 331(1):38-48. doi:10.1001/jama.2023.24945.
pmid: 38078870 |
| [4] | Cumpston M, Li T, Page MJ, et al. Updated guidance for trusted systematic reviews: A new edition of the cochrane handbook for systematic reviews of interventions[J]. Cochrane Database Syst Rev, 2019, 10(10):ED000142. doi:10.1002/14651858.ED000142. |
| [5] |
Ludvik B, Giorgino F, Jódar E, et al. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): A randomised, open-label, parallel-group, phase 3 trial[J]. Lancet, 2021, 398(10300):583-598. doi:10.1016/S0140-6736(21)01443-4.
pmid: 34370970 |
| [6] |
Dahl D, Onishi Y, Norwood P, et al. Effect of subcutaneous tirzepatide vs placebo added to titrated insulin glargine on glycemic control in patients with type 2 diabetes: The SURPASS-5 randomized clinical trial[J]. JAMA, 2022, 327(6):534-545. doi:10.1001/jama.2022.0078.
pmid: 35133415 |
| [7] | Inagaki N, Takeuchi M, Oura T, et al. Efficacy and safety of tirzepatide monotherapy compared with dulaglutide in Japanese patients with type 2 diabetes (SURPASS J-mono): A double-blind, multicentre, randomised, phase 3 trial[J]. Lancet Diabetes Endocrinol, 2022, 10(9):623-633. doi:10.1016/S2213-8587(22)00188-7. |
| [8] | Garvey WT, Frias JP, Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): A double-blind, randomised, multicentre, placebo-controlled, phase 3 trial[J]. Lancet (London, England), 2023, 402(10402):613-626. doi:10.1016/S0140-6736(23)01200-X. |
| [9] |
Rosenstock J, Frías JP, Rodbard HW, et al. Tirzepatide vs insulin lispro added to basal insulin in type 2 diabetes: The SURPASS-6 randomized clinical trial[J]. JAMA, 2023, 330(17):1631-1640. doi:10.1001/jama.2023.20294.
pmid: 37786396 |
| [10] | Samms RJ, Coghlan MP, Sloop KW. How may GIP enhance the therapeutic efficacy of GLP-1?[J]. Trends Endocrinol Metab, 2020, 31(6):410-421. doi:10.1016/j.tem.2020.02.006. |
| [11] | Stemmer K, Finan B, DiMarchi RD, et al. Insights into incretin-based therapies for treatment of diabetic dyslipidemia[J]. Adv Drug Deliv Rev, 2020, 159:34-53. doi:10.1016/j.addr.2020.05.008. |
| [12] |
Gao L, Lee BW, Chawla M, et al. Tirzepatide versus insulin glargine as second-line or third-line therapy in type 2 diabetes in the Asia-Pacific region: The SURPASS-AP-Combo trial[J]. Nature Medicine, 2023, 29(6):1500-1510. doi:10.1038/s41591-023-02344-1.
pmid: 37231074 |
| [13] | Kadowaki Takashi, Chin Rina, Ozeki Akichika, et al. Safety and efficacy of tirzepatide as an add-on to single oral antihyperglycaemic medication in patients with type 2 diabetes in Japan (SURPASS J-combo): A multicentre, randomised, open-label, parallel-group, phase 3 trial[J] .Lancet Diabetes Endocrinol, 2022, 10: 634-644. doi:10.1016/S2213-8587(22)00187-5. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||